
How Liposomal Glutathione Powder Protects Cells from Stress?
2026-04-27 15:37:39
Liposomal Glutathione Powder saves cells from oxidative stress by using a high-tech delivery method to wrap reduced L-glutathione in two layers of phospholipids. This mix protects the antioxidant from being broken down by stomach enzymes, so it can be taken directly into cells. Free radicals and reactive oxygen species damage DNA, proteins, and lipid layers in cells. Glutathione neutralizes these harmful substances once it is consumed. The liposomal encapsulation makes the glutathione pills 3–5 times more bioavailable than regular ones. This makes sure that enough antioxidant molecules get to the right tissues to effectively fight stress-induced cell harm.
Understanding Liposomal Glutathione Powder and Cellular Stress
Modern nutraceutical preparations need ingredients that don't have the same absorption problems as older ones. Glutathione is the body's main antioxidant, but taking supplements by mouth has traditionally been hard to absorb. Because of this, new transportation methods have been created to protect and move this important molecule.
What Makes Liposomal Technology Different
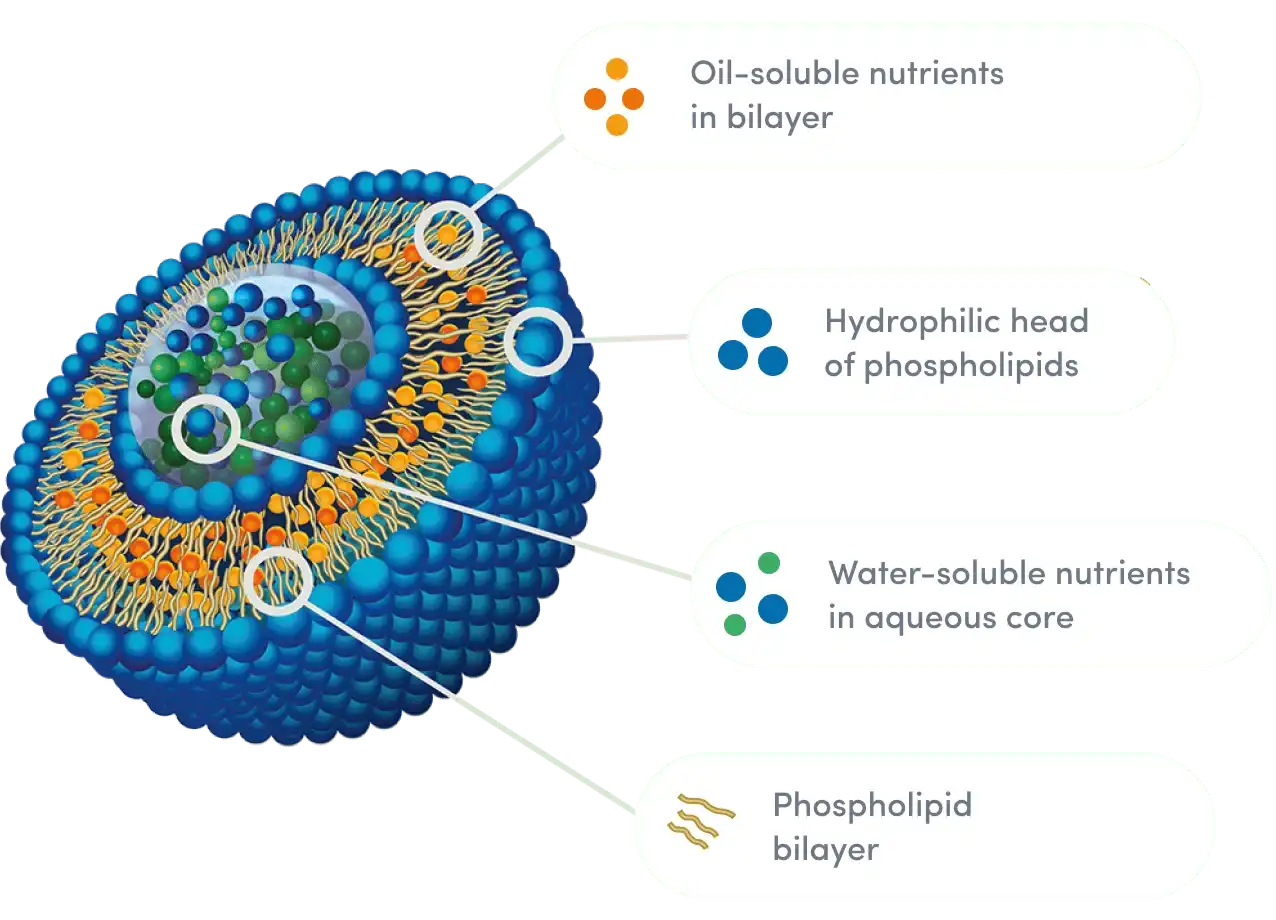
Liposome encapsulation is a way to give nutrients that is safe enough for use in pharmaceuticals. Glutathione molecules are wrapped up in tiny spheres made from phospholipids, which are the same stuff that makes up the walls of human cells. At Hongda, we make these protected structures with non-GMO sunflower lecithin and high-pressure homogenization. This makes sure that the particles are spread out evenly, which keeps the glutathione stable during digestion. Specifically, gamma-glutamyl transferase and stomach acid attack and break down glutathione quickly when it is not covered by the phospholipid membrane. This defense system lets the antioxidant that is enclosed get through the tough environment of the stomach and into the bloodstream straight through cells in the intestines. Then, our spray-drying method turns the liquid mixture into a stable powder. This stops the liposomes from breaking down because of wetness while keeping their structure.
The Cellular Stress Challenge
When reactive oxygen species build up faster than the body's antioxidant mechanisms can get rid of them, oxidative stress happens. These harmful chemicals are made by metabolic processes, inflammatory reactions, and environmental toxins. ROS affect any part of a cell, oxidizing membrane lipids, changing DNA patterns, and breaking down proteins that do their job. This chain reaction of damage speeds up aging, weakens the immune system, and makes chronic diseases more likely to happen. The amount of cellular glutathione determines how well cells can fight off this oxidative attack. This is why bioavailable supplements are strategically Liposomal Glutathione Powder important for health product developers who want to target markets for stress resistance and longevity.
Key Benefits of Liposomal Glutathione Powder in Stress Protection
Glutathione is a healing substance that affects many body systems. This makes it a useful addition for many different products. Liposomal Glutathione Powder in its encapsulated version makes these benefits even stronger by delivering them more efficiently to cells.
Neutralizing Free Radicals at the Source
Glutathione is an antioxidant that works on its own and with glutathione peroxidase enzymes. Hydrogen peroxide and lipid peroxides are neutralized by this two-step process before they start oxidative chain reactions. In the mitochondria of cells, where metabolic action creates the most ROS, glutathione keeps the redox balance that is needed to make energy. Liposomal delivery improves uptake, making sure that enough glutathione gets to these parts inside cells. This protects mitochondrial DNA and keeps cells' energy levels up when metabolic stress happens.
Supporting Skin Health and Anti-Aging Applications
The cosmetics business likes glutathione because it stops melanin from working. Glutathione lowers melanin production by stopping the oxidative stimulation of the tyrosinase enzyme. This helps with discoloration and uneven skin tone. Glutathione does more than just control color. It also keeps collagen and elastin fibers from oxidative cross-linking, which causes wrinkles and loss of skin flexibility. Our powder recipe mixes easily with vitamin C and other beauty-from-within ingredients, making synergistic formulas where the liposomal layer stops ingredients from reacting too soon in the dry blend phase. Because of this steadiness, companies can make high-quality capsules and beverage sachets with extended shelf life and reliable efficacy.

Liver Detoxification and Immune Modulation
The amount of glutathione in the liver directly affects its ability to detoxify. In order to get rid of toxins, heavy metals, and drug byproducts, the liver neutralizes them through glutathione conjugation. Hepatic glutathione stocks are depleted by drinking alcohol, having a fatty liver, and being exposed to toxins in the environment. This makes detoxification routes less effective. Taking supplements with highly bioavailable forms helps phase II detoxifying enzymes by replenishing these stores. Immune cells maintain high glutathione concentrations so that they can keep multiplying and making antibodies. Lymphocytes that are under reactive stress don't work as well, so the level of glutathione affects how well the immune system responds to infections or chronic inflammation.
Comparison and Procurement Insights: Choosing the Right Liposomal Glutathione Powder
When making a B2B procurement choice, you have to look at a lot of technical and business factors to make sure that the quality of the ingredients matches the positioning of the final product and meets legal standards. Liposomal Glutathione Powder offers measurable advantages over traditional forms.
Bioavailability Advantages Over Standard Forms
Traditional reduced glutathione powders suffer from poor oral absorption rates; often, below 15% of the dose given usually gets into the body's blood. Tablets and capsules with glutathione that are not enclosed are quickly broken down by enzymes in the small intestine, which limits their effectiveness. In clinical tests that compare liposomal and normal glutathione formulations, plasma glutathione levels are much higher after liposomal administration. This edge in bioavailability Liposomal Glutathione Powder means lower effective doses, lower raw material costs per serving, and a better way to set your product apart in a crowded market.
Quality Certifications and Sourcing Standards
For foreign trade in nutraceuticals, following the rules is a must. Hongda maintains cGMP approval, which makes sure that its production processes meet pharmaceutical-grade standards for keeping products clean, making sure that batches are consistent, and keeping track of them. Our SGS-certified laboratory uses HPLC to check the amount of glutathione and TEM imaging to confirm that the structure of the liposomes is correct. According to USP and EP guidelines, each production batch is checked for heavy metals, microbial contamination, and residual solvents. We are certified as HALAL, KOSHER, and ORGANIC, and hold FDA registration and BRC certification.

Practical Procurement Considerations
Minimum order amounts affect both the original investment and the management of inventory. Our 1kg MOQ allows for both test runs for new products and regular production runs, making it easier for new brands to see if the market will accept them. The product can be stored at room temperature for 24 months, which cuts down on the costs of cold chain operations and simplifies international shipping. Higher concentrations, like 70%, work better for compact dosage forms, while lower concentrations serve beverage applications. Long-term supply deals take advantage of our 3,000-ton annual production capacity across ten specialized manufacturing lines.
Safe Usage and Dosage Recommendations for Liposomal Glutathione Powder
Dosing standards based on evidence help formulators make safe, efficient products that meet the health needs of consumers and stay in line with regulations. Liposomal Glutathione Powder integration should follow clinical findings.
Clinical Dosage Ranges
Oral liposomal glutathione supplements have been studied and found to work at amounts ranging from 250 mg to 1,000 mg per day, based on the treatment goal. Applications that lighten the skin usually use 500 mg daily doses for 12 weeks. Formulations that help the liver may suggest taking 600 to 800 mg every day during cleansing programs. A lot of athletic healing items suggest 1,000 mg after working out to deal with the acute oxidative stress that comes from hard training. These amounts provide starting points for formulation development.
Safety Profile and Contraindications
Glutathione is very safe in clinical tests. At high doses, the most common side effect noted was mild stomach problems. The encapsulated form has a less sulfurous taste and smell, which makes it easier for people to take than versions that are not covered. People who are getting chemotherapy should talk to their doctors before taking supplements, because glutathione might affect some oxidative cancer treatments. Women who are pregnant or nursing should follow normal safety rules. No major drug interactions have been documented, but people who take immunosuppressant drugs should talk to a doctor.
Storage and Handling Best Practices
Maintaining product purity throughout the supply chain requires attention to environmental factors. Unlike liquid liposomal goods, which need to be kept cold, our powder version can be stored at room temperature. The main risk of degradation is being exposed to moisture. Using moisture-barrier materials and desiccant sachets in the package keeps the product's effectiveness throughout the shelf life. Light-resistant cases keep the phospholipid membrane from breaking down due to oxidation. When moving large amounts, sealed packages should be kept in climate-controlled stores where the humidity is less than 60%.
How to Integrate Liposomal Glutathione Powder into Your Product Line
To successfully integrate products, you need to know about Liposomal Glutathione Powder technology compatibility, regulatory routes, and supplier partnership factors that help your business grow in the long run. Liposomal Glutathione Powder offers exceptional versatility across delivery systems.
Formulation Strategies for Maximum Efficacy
The powder shape is very flexible when it comes to delivery methods. The powder's ability to flow well helps capsule formulas, allowing for automated encapsulation without problems with bridging. With the right additives, tablet compression remains possible, but capsules are better at keeping the liposomal structure. When the powder is mixed with water, it dissolves quickly and forms liposomal structures again. When you mix glutathione with vitamin C, you get stronger antioxidant protection. When you mix it with alpha-lipoic acid, you help cells recycle glutathione. Its neutral flavor profile permits incorporation into protein powders and functional coffee products.
Regulatory Compliance Across Global Markets
Dietary supplement rules in the US, set by the DSHEA, require manufacturers to follow cGMP guidelines and use honest labels. Any health claims must be backed up by reliable scientific proof, or they must be structure-function claims with the right disclaimers. EU Novel Food regulations generally permit glutathione supplementation, though some member states may have unique notice rules. Our complete documentation packages—including certificates of analysis, allergen statements, and manufacturing process descriptions—facilitate regulatory submissions and customs clearance. We stay up to date on how laws change in key export markets to help our clients with compliance.
Selecting a Reliable Manufacturing Partner
Supplier selection determines product quality, consistency, and business continuity. Being open about manufacturing processes shows that our operations are mature. We welcome client facility audits and maintain open communication regarding production schedules and quality measures. The ability to provide technical help is what sets strategic partners apart. Our research and development team, made up of professors from leading universities, works with clients to create unique formulations and plan clinical studies. Hongda's 23-year history in botanical extract manufacturing demonstrates stability and expertise that help clients navigate the tough nutraceutical market.
Conclusion
Cellular protection against oxidative stress is an important health goal for many consumer demographics. Liposomal Glutathione Powder gets around the problems with absorption that have historically constrained oral glutathione supplements, delivering clinically meaningful antioxidant support through advanced encapsulation technology. For B2B procurement specialists and brand developers, this ingredient offers differentiation in premium categories where scientific proof and efficacy drive purchasing decisions. Successful integration requires attention to formulation compatibility, regulatory compliance, and supplier capabilities.
FAQ1. What distinguishes liposomal glutathione from regular glutathione supplements?
The encapsulation method keeps stomach enzymes from breaking down glutathione, which makes absorption rates much higher. Standard glutathione powders are broken down before they get into the bloodstream. Liposomal forms, on the other hand, keep their molecular integrity through the digestive system.
2. Can liposomal glutathione powder be used in beverage formulations?
Our powder mixes well with water and forms liposomal structures again when it gets wet. It can be used in functional drinks, stick packs, and ready-to-mix supplements without having to hide the flavor too much because it has a neutral taste and no sulfurous smell.
3. What quality certifications should B2B buyers prioritize?
cGMP approval ensures processing meets standards for pharmaceuticals. Verification testing by SGS or similar labs shows that the ingredients are pure and effective. Certain market standards, like HALAL, KOSHER, and ORGANIC, make it easier for businesses to reach their target customers and demonstrate comprehensive quality management.
4. How does storage stability compare to liquid liposomal products?
Our powder recipe stays effective for 24 months at room temperature, eliminating costs associated with cold chain logistics. Liquid liposomal products typically require refrigeration and have shorter shelf lives due to moisture-catalyzed degradation and oxidation risks inherent in aqueous formulations.
Partner with a Trusted Liposomal Glutathione Powder Manufacturer
Hongda has been working on advanced liposomal formulations for more than twenty years and has a lot of experience with phytochemicals. Our 20,000-square-meter cGMP plant produces uniform, high-quality Liposomal Glutathione Powder backed by Certifications including FDA registration, ISO22000, and FSSC22000. We support global nutraceutical brands with flexible MOQs, custom specification creation, and quick expert support. Whether you're launching a new product line or optimizing existing formulations, our team provides the collaborative partnership necessary for success. Contact Us at duke@hongdaherb.com to discuss your specific requirements and receive detailed product specifications.
References
1. Sekhar, R.V., McKay, S.V., Patel, S.G., Guthikonda, A.P., Reddy, V.T., Balasubramanyam, A., & Jahoor, F. (2011). Glutathione synthesis is diminished in patients with uncontrolled diabetes and restored by dietary supplementation with cysteine and glycine. Diabetes Care, 34(1), 162-167.
2. Richie, J.P., Nichenametla, S., Neidig, W., Calcagnotto, A., Haley, J.S., Schell, T.D., & Muscat, J.E. (2015). Randomized controlled trial of oral glutathione supplementation on body stores of glutathione. European Journal of Nutrition, 54(2), 251-263.
3. Watanabe, F., Hashizume, E., Chan, G.P., & Kamimura, A. (2014). Skin-whitening and skin-condition-improving effects of topical oxidized glutathione: A double-blind and placebo-controlled clinical trial in healthy women. Clinical, Cosmetic and Investigational Dermatology, 7, 267-274.
4. Sinha, R., Sinha, I., Calcagnotto, A., Trushin, N., Haley, J.S., Schell, T.D., & Richie, J.P. (2018). Oral supplementation with liposomal glutathione elevates body stores of glutathione and markers of immune function. European Journal of Clinical Nutrition, 72(1), 105-111.
5. Allen, J., & Bradley, R.D. (2011). Effects of oral glutathione supplementation on systemic oxidative stress biomarkers in human volunteers. Journal of Alternative and Complementary Medicine, 17(9), 827-833.
6. Schmitt, B., Vicenzi, M., Garrel, C., & Denis, F.M. (2015). Effects of N-acetylcysteine, oral glutathione (GSH), and a novel sublingual form of GSH on oxidative stress markers: A comparative crossover study. Redox Biology, 6, 198-205.














