Liposomal Berberine Powder
2.Main Ingredients:Berberine
3.Specification:10%-90%(specification and process can be customized)
4.Assay:HPLC
5.Appearance:Yellow powder
6.Place of Origin:Shaanxi China
7.MOQ:1kg
8.Shelf Life:24 months
9.Product Source:Berberine
Liposomal Berberine Powder - Advanced Bioavailability Solution
As a driving producer and provider, Shaanxi Hongda Phytochemistry specializes in creating high-quality liposomal berberine powder through progressed epitome innovation. Our item conveys 5-10x improved bioavailability compared to standard berberine hydrochloride, with embodiment productivity >85% confirmed by HPLC investigation. The phospholipid bilayer framework bypasses P-glycoprotein efflux pumps, accomplishing molecule sizes of 100-400nm by means of Energetic Light Scrambling approval. This disposes of the gastrointestinal trouble and severe taste related with routine berberine. You advantage from our 20,000 square meter office, 100,000-level refinement workshops, and SGS standardized research facility guaranteeing reliable quality for your formulations.

Key Advantages
Enhanced Bioavailability
The liposomal conveyance framework increments assimilation rates by 5-10 times. Your clients encounter superior comes about with lower doses.
Improved Tolerability
Our epitome innovation disposes of the gastrointestinal side impacts common with standard berberine. This diminishes client complaints and moves forward satisfaction.
Taste Masking Properties
The phospholipid coating covers berberine's common sharpness. You can define pleasant-tasting items without extra flavoring agents.
Versatile Applications
The water-dispersible powder coordinating effectively into capsules, tablets, and useful refreshments. This adaptability bolsters differing item improvement strategies.
Liposomal Berberine Powder Specifications
|
Item
|
Specification Method
|
Result
|
Method
|
|
Physical and Chemical Property
|
|||
|
Appearance
|
Yellow Fine Powder
|
Conforms
|
Visual
|
|
Particle Size
|
95% through 80 mesh
|
Conforms
|
Screening
|
|
Residue on Ignition
|
≤5g/100g
|
1.28g/100g
|
3g/550℃/4hrs
|
|
Loss on Drying
|
≤5g/100g
|
3.05g/100g
|
3g/105℃/2hrs
|
|
Identification
|
|
Conforms
|
TLC
|
|
Content:
|
Liposomal Berberine>30%
|
Conforms
|
HPLC
|
|
Residue Analysis
|
|||
|
Heavy Metals
|
≤10.0mg/kg
|
Conforms
|
|
|
Lead (Pb)
|
≤2.00mg/kg
|
Conforms
|
ICP-MS
|
|
Arsenic (As)
|
≤2.00mg/kg
|
Conforms
|
ICP-MS
|
|
Cadmium (Cd)
|
≤1.00mg/kg
|
Conforms
|
ICP-MS
|
|
Mercury (Hg)
|
≤0.01mg/kg
|
Conforms
|
ICP-MS
|
|
Microbiological Tests
|
|||
|
Total Plate Count
|
≤1000cfu/g
|
124cfu/g
|
AOAC 990.12
|
|
Total Yeast & Mold
|
≤100cfu/g
|
45cfu/g
|
AOAC 997.02
|
|
E.Coli.
|
Negative/10g
|
Conforms
|
AOAC 991.14
|
|
Salmonella
|
Negative/10g
|
Conforms
|
AOAC 998.09
|
|
S.aureus
|
Negative/10g
|
Conforms
|
AOAC 2003.07
|
|
Product Status
|
|||
|
Conclusion:
|
Sample Qualified.
|
||
|
Shelf Life:
|
24 months under the conditions below and in its original packaging.
|
||
|
Retest date:
|
Retest every 24 months under the conditions below and in its original packaging.
|
||
|
Storage:
|
Store in a cool, dry place away from Moisture, Light.
|
||
Manufacturing Process
Our progressed generation handle guarantees prevalent quality and bioavailability:
Step 1: Lipid Preparation
We plan pharmaceutical-grade phospholipid bilayers utilizing non-GMO sunflower phosphatidylcholine. The lipids experience quality testing for virtue and composition.
Step 2: Berberine Encapsulation
Through controlled temperature blending and high-pressure homogenization, berberine is entangled inside liposome structures. This makes the defensive lipid barrier.
Step 3: Size Optimization
The liposome suspension experiences sonication and expulsion to accomplish uniform molecule estimate dispersion between 100-400nm for ideal cellular uptake.
Step 4: Lyophilization
Advanced freeze-drying innovation changes over fluid liposomes into steady powder shape whereas keeping up basic astuteness and bioactivity.
Step 5: Quality Verification
Each clump experiences comprehensive testing counting embodiment proficiency, molecule estimate investigation, and TEM imaging to affirm liposome formation.
Applications Dietary Supplements
High-bioavailability metabolic bolster details focusing on blood glucose direction and lipid administration. The improved retention permits for "low-dose, high-effect" item positioning.
Functional Foods
Water-dispersible properties make it perfect for powdered drink blends, smoothie definitions, and useful sachets where conventional berberine would make agreeability issues.
Clinical Nutrition
Medical nourishment applications for PCOS and metabolic disorder administration require reliable plasma levels. Our liposomal framework conveys dependable bioavailability for helpful outcomes.
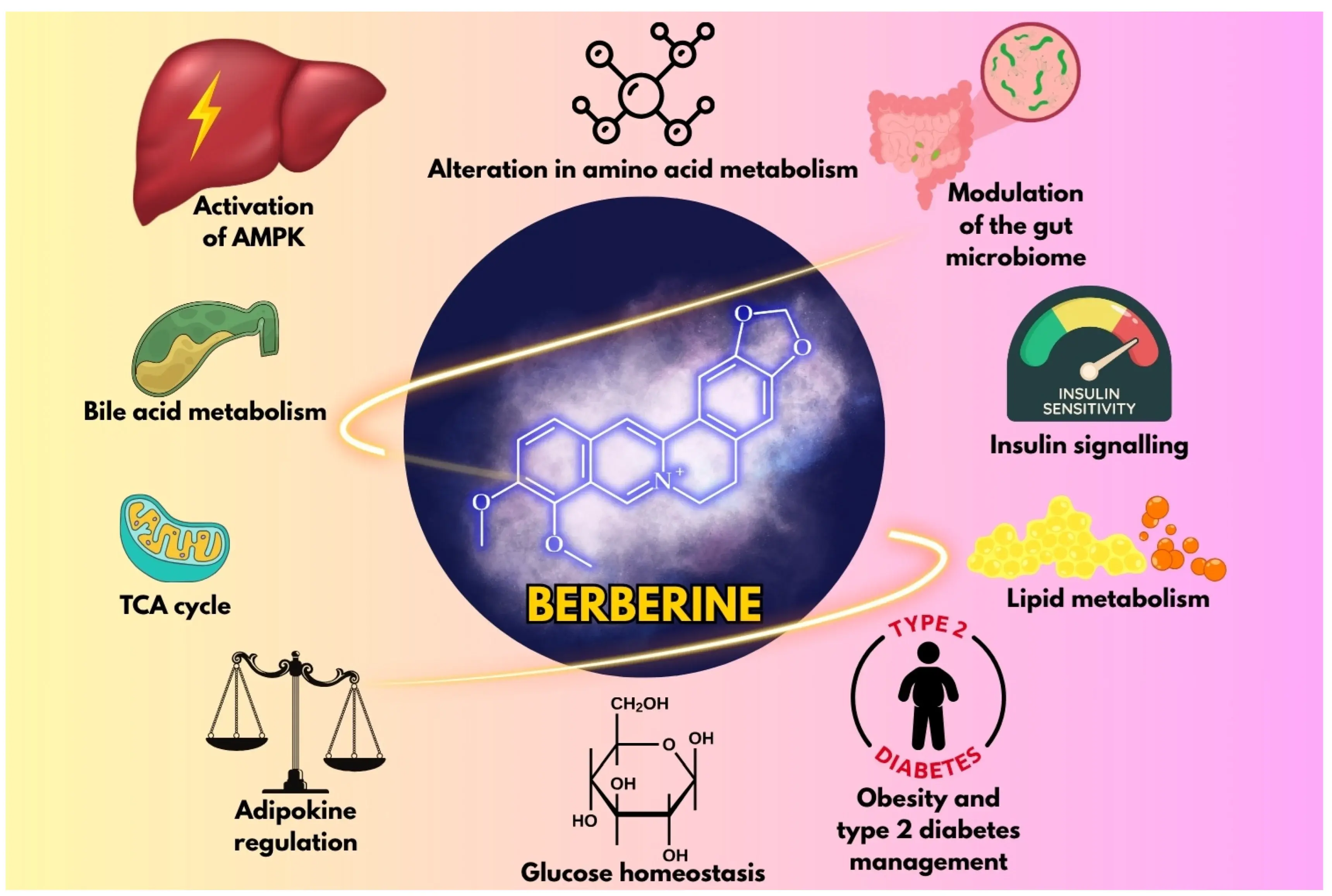
Quality Assurance
Our comprehensive quality control framework approves item realness and safety:
Encapsulation Efficiency Testing
Ultracentrifugation taken after by HPLC examination affirms >85% berberine entanglement, recognizing true liposomes from straightforward physical mixtures.
Particle Characterization
Dynamic Light Scrambling (DLS) estimations confirm nanometric estimate run and zeta potential showing electrostatic stability.
Morphology Verification
Transmission Electron Microscopy (TEM) gives visual affirmation of round bilayer structures or maybe than nebulous aggregates.
Purity Analysis
HPLC evaluation combined with overwhelming metal testing (Pb, As, Cd, Hg) and remaining dissolvable investigation guarantees compliance with worldwide standards.

Why Choose Hongda?(liposomal berberine manufacturer)
Advanced Manufacturing
Our 3,000-ton yearly capacity office highlights present day generation lines and 100,000-level refinement workshops. Over 150 talented specialists guarantee reliable quality.
Research Excellence
More than 20 professor-level R&D work force work in our SGS standardized research facility. College associations drive nonstop advancement in liposomal technology.

Quality Certifications
We keep up HALAL, Legitimate, cGMP, FSSC22000, ISO9001, and ISO22000 Certifications. These qualifications back your administrative prerequisites around the world.

Custom Solutions
Our OEM/ODM capabilities allow specification customization from 10-90% concentration. We adapt formulations to meet your specific market needs.

FAQ
Q: How does bioavailability compare to standard berberine?
A: Pharmacokinetic studies show 5-10x increases in Cmax and AUC because liposomes bypass P-gp efflux pumps and first-pass hepatic metabolism.
Q: Is this true liposome or just phospholipid blend?
A: This is verified true liposome. We provide Encapsulation Efficiency data and TEM imagery proving berberine entrapment within lipid bilayers.
Q: Does the powder contain excipients?
A: Yes, stabilizing matrices like maltodextrin ensure powder stability during drying. Active berberine load is titrated to specific percentages for dosing accuracy.
Q: Is the powder water-soluble?
A: It's water-dispersible. Upon mixing with water, powder rehydrates into stable liposomal suspension suitable for liquid applications.
Q: What about shelf life and stability?
A: Liposomal powders resist oxidation and hydrolysis better than liquid forms. Proper storage below 25°C provides 24-month shelf life.
Contact us
Ready to enhance your product line with superior liposomal berberine powder? Contact our technical team at duke@hongdaherb.com for detailed specifications and samples.



_1772003989774.jpg)
_1772003990646.jpg)













