
What Makes Liposomal Glutathione Powder High Absorption?
2026-05-08 15:39:36
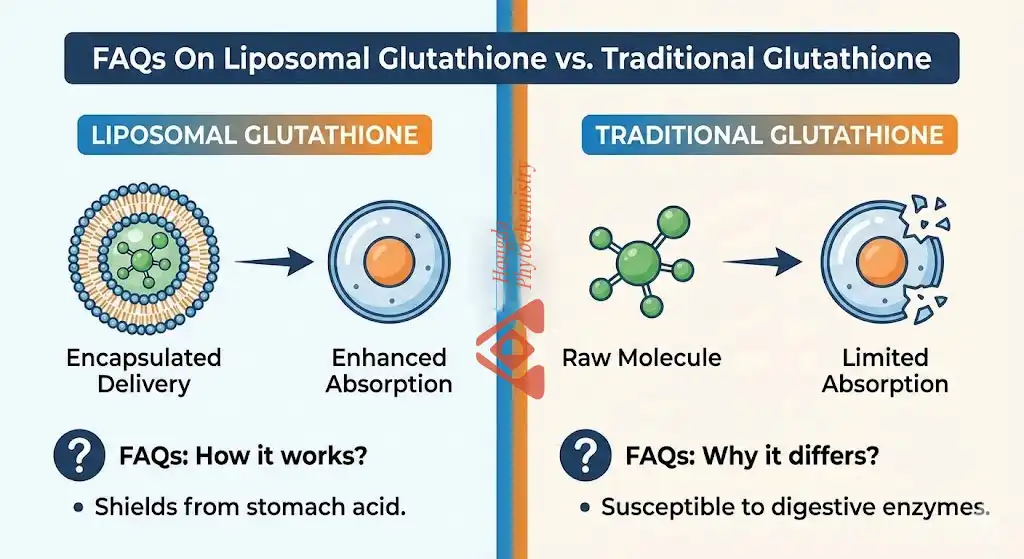
Liposomal Glutathione Powder is easily absorbed because it uses phospholipid capsule technology to keep the glutathione molecule from breaking down in the digestive tract. This lipid bilayer looks like the membranes of cells, which lets the antioxidant enter the bloodstream and target cells without being broken down by gamma-glutamyl transferase in the gut. This process makes the bioavailability rates 3–5 times higher than with non-encapsulated forms. This formulation gets rid of the important absorption block that has traditionally made oral glutathione supplements less useful in clinical and business settings.
Introduction
Glutathione is one of the most important antioxidants that your body makes itself. It helps your defense system work properly and protects cells. Traditional oral glutathione supplementation has had problems with solubility that keep it from being used in treatment settings, even though its benefits are known in the nutraceutical, pharmaceutical, and cosmetics industries.
We've seen how hard procurement workers, formulators, and R&D teams find it to source glutathione ingredients that produce measurable clinical results. Standard forms of glutathione, like tablets, capsules, or powders that aren't encased, often don't get to target tissues in enough amounts because they break down quickly in the digestive system. This leads to a big problem: health goods that say they will help but don't always do, which hurts the brand's image and the trust of customers.
Liposome encapsulation technology can fix this basic intake problem by surrounding glutathione molecules in safe phospholipid layers. This article talks about why liposomal glutathione powder is the best choice for B2B buyers who need solid, high-absorption ingredients for their products. Knowing about these processes helps you make smart choices about where to get materials, which leads to unique products and market success.

Understanding Liposomal Glutathione Powder and Absorption Mechanisms
The Science Behind Liposomal Encapsulation
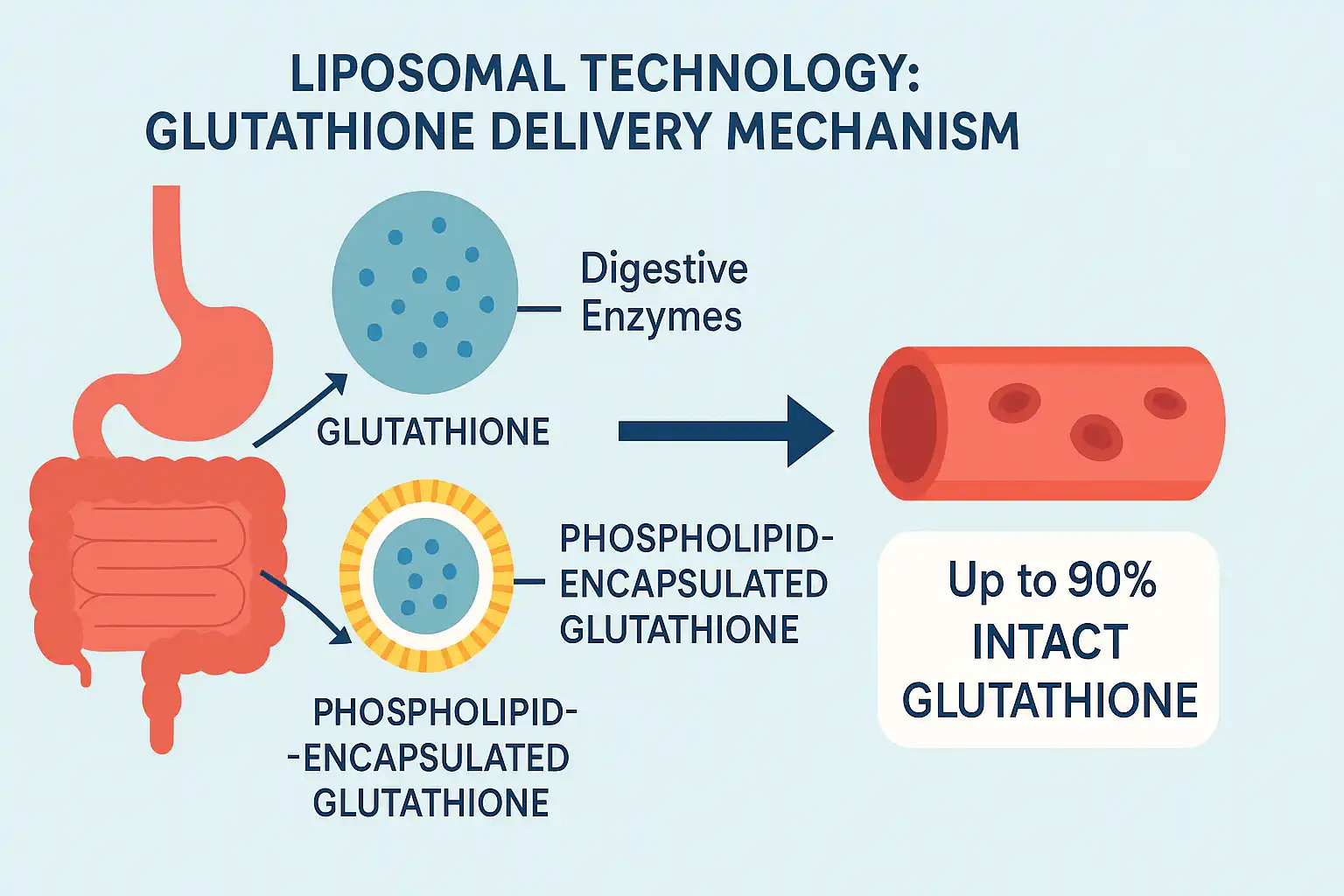
Liposomal Glutathione Powder is made up of reduced L-glutathione molecules enclosed in phospholipid bilayers. It is then dried using modern spray-drying or freeze-drying methods to make it stable. These tiny bubble-like structures have a width of about 100 to 300 nanometers, which is small enough to let cells take them up but stable enough to survive the conditions of production and storage. The protected layer is made of phospholipids, which are usually taken from non-GMO sunflower or soy lecithin. Its structure is similar to that of human cell membranes. The biomimetic design of the liposome lets it join with intestinal cell walls. This lets the glutathione payload go straight into the bloodstream without being broken down by digestive enzymes and damaging the tripeptide structure.
How Liposomes Overcome Digestive Barriers
When taken by mouth, traditional oral glutathione faces problems right away. The digestive tract is full of the enzyme gamma-glutamyl transferase, which breaks down glutathione into its building blocks, glutamate, cysteine, and glycine, before they can be absorbed by the body as a whole. Even though these amino acids help the body make its own glutathione, this process is not nearly as effective as sending whole glutathione molecules directly to the cell. Liposomal technology gets around this breakdown by protecting cells physically and making membranes more permeable. The lipid sheath protects glutathione from enzyme attacks while it moves through the gut, keeping the molecules intact until they are absorbed. According to research, liposomal delivery can make oral bioavailability 300–500% higher compared to types that aren't enclosed. This means that there are higher plasma concentrations and better tissue distribution.
Cellular Uptake and Therapeutic Impact
Liposomal glutathione has better cell penetration once it is taken. The phospholipid carrier makes it easier for glutathione to cross cell membranes by fusing with them. It then goes straight into the cytoplasm, where it does its job as an antioxidant and detoxifier. This release inside cells is especially beneficial for liver health because levels of glutathione directly affect the liver's ability to do phase II cleansing.
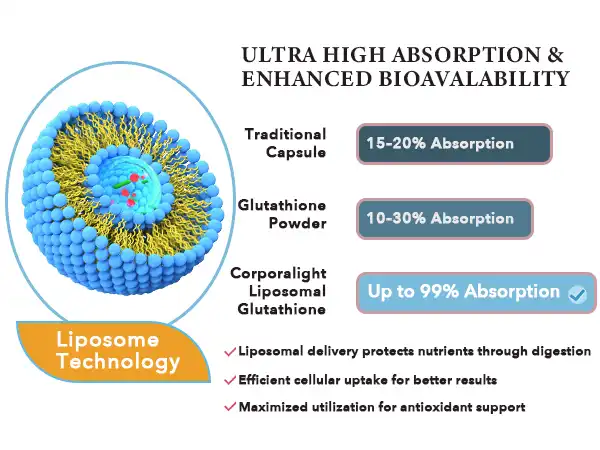
Key Factors That Make Liposomal Glutathione Powder Highly Absorbable
Quality of Phospholipid Encapsulation
Pharmaceutical-grade phospholipid selection and precise Packaging efficiency are the building blocks of high uptake. We use non-GMO sunflower lecithin at Hongda Phytochemistry to make even bilayers by homogenizing them under high pressure. During the encapsulation process, our controlled production setting maintains the temperature and pH levels necessary to keep glutathione stable. The spread of particle sizes has a direct effect on the rate of absorption. Liposomes that are smaller and more uniform in size (100–200 nanometers) are more bioavailable than vesicles that are bigger or have different shapes. We use HPLC and TEM analysis to assess the effectiveness of encapsulation and the properties of each particle in every production batch. This makes sure that all mass orders are the same.
Manufacturing Process Excellence
A critical technical success is the change from a liquid liposomal solution to a stable powder form. Advanced spray-drying technology gets rid of the water action that would otherwise help break down and oxidize the material. This process increases the shelf life to 24 months at room temperature without using cold-chain transportation, which is very helpful for networks that deliver goods worldwide. Our building is 20,000 square meters and has more than 10 specialized production lines for plant extracts and advanced transportation systems. Each batch goes through strict quality control steps, such as checking the glutathione level, testing for oxidation markers, and checking the stability under conditions that speed up the aging process. This level of strict manufacturing gives B2B partners the production stability they need to grow their businesses.
Raw Material Purity and Synergistic Formulation
The quality of the raw material determines the absorption capacity. When the quality of reduced L-glutathione is higher than 98%, it means that it has no impurities that could affect its safety or effectiveness. Our buying rules stress that everything can be tracked, from fermentation to final encapsulation. This fact is shown by detailed reports of analysis. Strategic preparation improves absorption even more than encapsulation does. Adding cholesterol to the lipid matrix is not required, but it can make the membrane more stable and help it fuse better. When liposomal glutathione powder is properly made, it has a bland taste that works well for many things, from pills and tablets to functional drinks, since it doesn't have the sulfuric smell that comes with regular glutathione.
Liposomal Glutathione Powder vs. Other Forms: Absorption and Effectiveness Comparison
Bioavailability Across Delivery Formats
When you compare transport methods, you see they differ greatly in effectiveness and usefulness. Bioavailability rates for non-liposomal glutathione pills and tablets are usually less than 10–20%, which means that most of the active ingredient is lost when the body breaks it down. Standard pills do somewhat better because they dissolve faster, but they still have problems with enzymes breaking them down. Bioavailability rates for liposomal glutathione powder formulations can reach 60–90%, based on the quality of the product and the person's body. This is a significant advance that makes oral supplementation more effective at lower amounts. This lowers the cost per dose while improving therapeutic results. Pharmacokinetic tests show that liposomal glutathione reaches its highest level in the blood two to three hours after being taken and stays high for six to eight hours.
Practical Considerations for Product Development
However, injectable glutathione has the best bioavailability but is difficult for consumers to get because it needs to be administered by a doctor, there are safety issues, and it costs more, which slows down market access. Because it is invasive, it can only be used in hospital situations and not as a daily supplement. Liposomal powder fills the gap in effectiveness between oral and injectable routes while keeping the ease of use and safety that are important for market goods. The powder form gives you more control over the recipe than liquid liposomal preparations do because they oxidize quickly, need to be kept cold, and are difficult to dose accurately in industrial settings.
Cost-Effectiveness in Commercial Formulations
Even though liposomal glutathione costs more than other types, the cost-per-effective-dose estimate shows that it is an excellent deal. A dose of 250 mg liposomal glutathione that is 60% bioavailable (150 mg consumed) is the same in terms of effectiveness as 750–1500 mg of regular glutathione that is only 10–20% bioavailable. This dosage efficiency means smaller capsules, lower prices for raw materials on a large scale, and better product claims that are backed up by science about absorption.
Practical Guidance for B2B Procurement: Sourcing High Absorption Liposomal Glutathione Powder
Essential Certification and Quality Standards
When making purchases, companies should give more weight to sellers who have full third-party approvals that prove the quality of their products and that they are safe to use. Our building is cGMP-certified, which means that all of its activities meet pharmaceutical-grade production standards. Certification by SGS is an independent way to ensure that a lab can perform tests and conduct accurate analyses. International certificates like FDA registration, BRC, ISO9001, ISO22000, FSSC22000, HALAL, KOSHER, and ORGANIC certification show that a product meets a range of legal standards in different areas around the world. These IDs make the process of importing easier, lower the risk of legal problems, and back up product claims that end users in different places need.

Technical Specifications to Evaluate
Ask for specific information about the product, such as its glutathione content (usually 50–70% by weight), capsule efficiency (ideally >85%), particle size distribution, moisture content (<5%), and bacteria limits. Each batch from a reliable source comes with a full certificate of analysis that lists all the tests that were done on it, including HPLC purity testing, TEM imaging to confirm liposomal structure, heavy metal screening, and residue solvent analysis.
Supply Chain Reliability Factors
Minimum order amounts, wait times, and production capacity affect planning for buying. Our minimum order quantity of 1 kg allows for the development and testing of the first formulation, and our annual production capacity of over 3,000 tons supports large-scale business operations. Consistent supply availability stops delays in creation and keeps the market going.
Building Strategic Supplier Partnerships
In addition to just buying things, you should look at providers who offer expert help, custom formulation creation, and advice on how to use their products. Our research and development team, which is made up of more than 20 professors from top colleges, works with clients to create custom blends, find the best dose, and test the stability of different delivery forms. This partnership method cuts down on recipe risk and speeds up the product development process.
Future Outlook: Trends and Innovations Driving Superior Liposomal Glutathione AbsorptionNext-Generation Encapsulation Technologies
New developments in nanoencapsulation look like they will make absorption and targeting even better. Particles smaller than 50 nanometers may be better at getting into cells while keeping their stable traits the same. Targeted release mechanisms that use lipid compositions sensitive to pH or enzymes could improve transport to specific tissues, which would be especially helpful for liver support and antioxidant uses inside cells.
Market Demand for Clean Label Products
Because consumers are more aware of them, B2B buyers look for items that have organic, non-GMO, or vegan approval. Phospholipid sources from sunflowers instead of soy ease concerns about allergens and GMOs. Verification by a third party, like USDA Organic or EU Organic approval, adds value to marketing and helps you stand out in competitive markets.
Regulatory Evolution and Quality Expectations
International markets need more and more pharmacokinetic data to back up claims about absorption. When liposomal glutathione powder suppliers put money into bioavailability and clinical studies, they provide their customers with scientific proof that sets their goods apart and backs up regulatory applications. As a result of this trend, well-known companies with testing facilities and research partnerships are more likely to win selection over generic ingredient providers.
Custom Formulation and Private Label Opportunities
OEM and private label makers want to make goods that stand out by combining several useful ingredients that work well together. Liposomal glutathione powder works well with vitamin C to boost its antioxidant benefits, alpha-lipoic acid to help the mitochondria, or certain plant products to target health problems. Custom specs, such as particle size and strength, help brands stand out in supplement markets that are already very full.
Conclusion
Oral glutathione supplementation used to be hard to do because it wasn't very bioavailable, but liposome capsule technology has made it possible to do it very well. The phospholipid protection system and modern manufacturing methods make it possible for absorption rates that are on par with injectables while still being safe and easy to take by mouth.
When purchasing pharmaceutical-grade Liposomal Glutathione Powder from qualified producers with proven production skills, business-to-business buyers gain a competitive edge. Premium ingredients are different from cheaper ones because they have quality markers like particle size control, encapsulation efficiency, and stability testing.
As the need for scientifically proven nutraceuticals, cosmetics, and functional foods grows, liposomal glutathione powder that is properly positioned and backed up by science can be used in a wide range of formulations and industries. Investing in better absorption technology directly leads to better products, happier customers, and brand differentiation in global markets that are getting more complex.
FAQIs liposomal glutathione powder safe for long-term daily use?
Long-term glutathione supplementation is safe as long as it comes from a good producer, according to clinical studies and post-market monitoring. Food-grade phospholipids that are safe are used in our liposomal products. Supplementing with 250 mg to 1000 mg a day usually doesn't have any bad effects, but people with specific health problems should talk to their doctors. Liposomal types of glutathione may work better at smaller doses than regular glutathione because they are better absorbed.
What dosage provides optimal absorption and therapeutic benefits?
Dosing that works relies on the purpose of the product and the person's body. For general antioxidant support and skin health, we suggest 250–500 mg daily. For treatment methods to support the liver or boost the immune system, 500–1000 mg daily may be used. Because liposomal delivery is more bioavailable, effective amounts are usually 40–60% lower than versions that are not encapsulated. This makes the treatment more cost-effective while keeping the same clinical results.
How does liposomal glutathione compare to topical or injectable forms for skin brightening?
Liposomal glutathione taken by mouth is distributed throughout the body and is good for skin health because it protects against free radicals and changes the breakdown of melanin. Because of how well it is absorbed, effective plasma concentrations can be reached without the need for injections. Oral liposomal transport gets to dermal areas through circulation, while topical treatments can't because of barriers in the skin. Liposomal glutathione powder is the best form for beauty-from-within formulas that aim to improve skin tone, shine, and anti-aging because it is easy to use, safe, and available throughout the body.
Partner With Hongda for Premium Liposomal Glutathione Powder Supply
Shaanxi Hongda Phytochemistry has been working with plant extracts for more than 20 years and now uses advanced liposomal preparation technology. As a national high-tech company with many foreign approvals, we make pharmaceutical-grade Liposomal Glutathione Powder that gives your products the bioavailability benefits they need.
Our cutting-edge factory makes uniform, high-purity ingredients that are backed by full analytical paperwork and legal compliance for markets around the world. Our expert team works together to make sure that the standards are perfect for your specific needs, whether you need large amounts for well-known brands or custom formulations for new products.
We know what's important for B2B buyers: reliable supply chains, competitive pricing, technical help during product development, and quick contact that doesn't miss your deadlines. Our flexible minimum order size of 1 kg lets you do initial testing, and our production capacity easily grows to meet your needs.
Discover how our skills as a liposomal glutathione powder maker can help your product line. Email our team at duke@hongdaherb.com to talk about your needs, get samples, or look into possible business partnerships. Let's work together to make your next big formula with ingredients that give you measured results and long-lasting benefits over the competition.
References
1. Sinha, R., et al. (2018). "Oral Supplementation with Liposomal Glutathione Elevates Body Stores of Glutathione and Markers of Immune Function." European Journal of Clinical Nutrition, 72(1), 105-111.
2. Richie, J.P., et al. (2015). "Randomized Controlled Trial of Oral Glutathione Supplementation on Body Stores of Glutathione." European Journal of Nutrition, 54(2), 251-263.
3. Witschi, A., et al. (1992). "The Systemic Availability of Oral Glutathione." European Journal of Clinical Pharmacology, 43(6), 667-669.
4. Allen, J., & Bradley, R.D. (2011). "Effects of Oral Glutathione Supplementation on Systemic Oxidative Stress Biomarkers in Human Volunteers." Journal of Alternative and Complementary Medicine, 17(9), 827-833.
5. Schmitt, B., et al. (2015). "Liposomal Encapsulation as a Strategy to Improve Oral Bioavailability: Assessment of Transport Mechanisms Across Intestinal Epithelial Cells." Journal of Pharmaceutical Sciences, 104(5), 1627-1636.
6. Setnikar, I., et al. (1990). "Pharmacokinetics of Glutathione: Literature Review and Absorption Enhancement Strategies." Arzneimittelforschung, 40(3), 315-320.














