
Best Uses of Liposomal NMN Powder in Supplement Formulas
2026-04-08 15:09:27
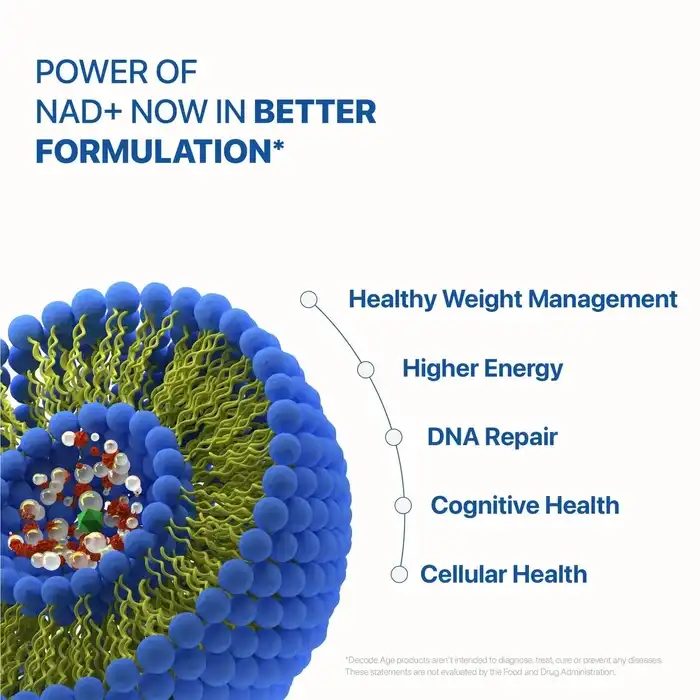
The pharmaceutical and nutraceutical industries increasingly recognize Liposomal NMN Powder as a revolutionary delivery system that addresses critical bioavailability challenges in NAD+ precursor supplementation. This advanced formulation technology represents a significant breakthrough for manufacturers seeking to create premium anti-aging and wellness products. By encapsulating Nicotinamide Mononucleotide within phospholipid bilayers, this innovative powder form delivers superior cellular uptake while maintaining stability throughout the manufacturing and storage processes, making it an ideal choice for sophisticated supplement formulations targeting discerning health-conscious consumers.
Understanding Liposomal NMN Powder and Its Advantages
Liposomal NMN Powder is different because it uses a high-tech phospholipid-based transport system that completely changes how the body uses this important NAD+ precursor. Unlike regular NMN supplements, which are broken down a lot in the digestive system, this encapsulated form uses sunflower phospholipids to surround each NMN molecule with safe lipid spheres.
Enhanced Visualization Through Advanced Encapsulation
When it comes to cell uptake, the capsule technology used in high-quality liposomal products works really well. Researchers have found that liposomal delivery methods can make drugs up to 400% more bioavailable than regular powder forms. This improvement happens because the phospholipid shell is made to look like a cell membrane. This lets it directly fuse with intestinal cell walls and get around normal digestion problems.
Our production method at Shaanxi Hongda gets encapsulation efficiency of more than 85%, which means that the medicine has the most therapeutic potential. The particle size range of 100–300nm makes intestinal digestion better and keeps the product stable over its entire existence. The main problem with NMN is that it is naturally unstable in water. This precise engineering solves that problem.

Regulatory Compliance and Safety Profiles
Quality control is still very important when making pharmaceutical-grade supplements. Our Liposomal NMN Powder is certified by SGS and meets strict international standards, such as cGMP and FDA requirements. Each batch goes through a long list of tests, such as microbial analysis, heavy metal screening, and chemical residue identification.
The safety profile of liposomal NMN shows that it is well tolerated by a wide range of population groups. Clinical studies show that it has few negative effects when taken in the suggested dosage amounts. This means that it can be used as a supplement for a long time. When stored properly, the 24-month shelf life gives producers more options for managing their supplies and getting their products to customers.
Key Benefits of Incorporating Liposomal NMN Powder in Supplement Formulas
When liposomal NMN technology is added to supplement formulas, it gives measured benefits that lead directly to happier customers and more market differentiation. The solid evidence for these benefits keeps growing thanks to ongoing study projects around the world.
Cellular Energy Enhancement and Anti-Aging Applications
NAD+ production is the most important step in how cells use energy and how they stay healthy. Liposomal NMN Powder helps this process work well by sending whole NMN molecules straight to the parts of cells where they can be converted to NAD+ most efficiently. The rate-limiting factors in normal absorption routes are skipped by this tailored delivery system.
Studies show that constant NAD+ increase through optimal NMN supplementation is linked to better mitochondrial function, better DNA repair mechanisms, and activation of sirtuins. These are proteins that are directly linked to maintaining cellular health and life. People who take supplements notice these physiological changes by having more energy, better brain function, and better physical performance.
Market Positioning and Consumer Appeal
People who care about their health and are willing to spend money on advanced wellness solutions really like how liposomal delivery systems are positioned as high-end. Market study shows that people are looking for scientifically-backed goods that work better than basic formulations more and more.
Adding liposomal technology to a product makes it possible to differentiate it, which lets makers charge higher prices while still providing real value. Sunflower phospholipids as a carrier system are a good choice for clean labels because they are natural and don't contain GMOs.
Comparing Liposomal NMN Powder with Alternative NMN Forms
Knowing how the different NMN delivery methods compare to each other helps procurement workers and formulators make smart choices. Each shipping method has its own pros and cons that affect how well the product works and where it stands in the market.
Stability and Shelf Life Considerations
It's hard for traditional NMN powders to stay stable, especially in wet places or when they're exposed to high temperatures. Because normal NMN is hygroscopic, it needs complex packing and controlled storage conditions to stay effective over the course of its lifecycle.
Liposomal NMN Powder gets around these problems by enclosing the active ingredient in a secure shell that keeps it safe from external factors that break it down. The freeze-drying method used in production makes a stable powder that stays structurally sound when stored in normal circumstances. This improved steadiness means less product loss, better inventory management, and more trust from customers in the quality of the products.
Cost-Effectiveness Analysis
Liposomal versions usually have higher raw material costs than regular NMN powders, but because they are more bioavailable, they need less of a dose to work. This optimization can help partly make up for the higher price while still providing better treatment results.
Powder types are better for manufacturing efficiency than liquid liposomal preparations because they are easier to handle, cost less to ship, and last longer on the shelf. The 1kg minimum order amount that Shaanxi Hongda offers makes it easy for small and medium-sized businesses to test market possibilities or create niche product lines.
Procurement Insights: Buying Liposomal NMN Powder for Bulk and OEM Use
To strategically buy liposomal NMN, you need to carefully look at the skills, quality systems, and stability of the manufacturers. Because making liposomes is so complicated, it needs specialized knowledge and high-tech tools that set qualified sellers apart from stock providers.
Supplier Qualification and Quality Assurance
Reputable Liposomal NMN Powder makers show that their products meet international pharmacy standards by showing a wide range of Certifications. cGMP approval, SGS laboratory recognition, and ISO quality management systems are all necessary skills. With these certificates, you can be sure that the manufacturing method and quality control measures are always the same.
Shaanxi Hongda has been making things for 20 years and has been recognized as a national high-tech company. This shows that they are very good at extracting natural products and making advanced delivery systems. Our team of more than 150 trained workers does their jobs in purification workshops that are 100,000 times cleaner. These workshops are equipped with high-pressure homogenization technology that makes sure that particle sizes are evenly distributed and that Packaging works well.
Pricing Structures and Volume Considerations
The market price for liposomal NMN represents the complex manufacturing methods and specialty tools needed for reliable production. Rates on the market today depend on how well the capsules are sealed, how pure they are, and the guidelines for approval. Volume-based price systems usually have big benefits for well-known companies that can predict how much demand there will be.
Strategies for negotiating should focus on building long-term partnerships instead of just doing business. Suppliers who care about their customers' success by offering expert help, the ability to make custom formulations, and quick contact go above and beyond just providing raw materials.

Best Practices for Formulating with Liposomal NMN Powder
To successfully add liposomal NMN to vitamin formulas, you need to know the best ways to dose it, how to keep it stable, and how to make sure it works with other ingredients. These things have a direct effect on how well the product works and how satisfied customers are with it.
Dosage Optimization and Administration Protocols
Based on clinical studies, Liposomal NMN Powder formulations seem to work better than other ways of delivering drugs at lower amounts. Because it is more bioavailable, dosages between 125 mg and 500 mg per day can work, based on the target population and the purpose of the treatment.
When it comes to getting the most out of absorption, timing is very important. Giving it in the morning on an empty stomach improves uptake rates and reduces the chance of interactions with food components that could damage liposome structure. Product labels should make it clear how to use the product correctly so that customers always have the same experience.
Formulation Stability and Packaging Requirements
To keep the purity of the liposomes during the production process, it is important to pay close attention to the processing conditions and make sure that the ingredients work well together. High-shear mixing, too much heat exposure, and excipients that don't work well together can all hurt the effectiveness of encapsulation.
Protecting against wetness and light should be a big part of packaging so that the product stays stable. Barrier packing materials and desiccant inclusions make medicines last longer while keeping their restorative effectiveness. For unique uses, our expert team offers full formulation support to improve stability profiles and manufacturing efficiency.
Conclusion
Integrating Liposomal NMN Powder into supplement formulas is a big step forward in the fields of anti-aging and cellular health supplements. This advanced transport technology solves basic bioavailability problems and gives makers a premium ingredient base to set themselves apart in the market. Liposomal NMN is an important part of forward-thinking supplement brands that want to take the lead in the fast-growing life market because it has better therapeutic efficacy, better stability profiles, and a lot of customer appeal.
FAQWhat makes liposomal NMN more effective than standard NMN supplements?
Liposomal coating keeps NMN molecules from breaking down in the stomach and improves cell uptake through membrane fusion processes. This makes bioavailability and treatment effectiveness much better than with traditional delivery methods.
How should liposomal NMN powder be stored to maintain potency?
Keep away from direct light and moisture, and keep in a cool, dry place. Liposomal NMN powder stays active for 24 months if it is kept properly. Long-term stability is best achieved with desiccant packing and temperature-controlled settings.
What certifications should I look for when I buy liposomal NMN powder?
Some important certifications are cGMP production compliance, SGS laboratory approval, and any necessary organic certifications. ISO standards, HALAL/KOSHER certification, and FDA registration for entry to foreign markets are some other signs of quality.
Partner with Hongda for Premium Liposomal NMN Solutions
Hongda's advanced liposomal NMN powder manufacturing skills give supplement brands around the world unbeatable quality and stability. Our cutting-edge factories make pharmaceutical-grade ingredients that have been shown to encapsulate more than 85% of the time. We back this up with a lot of technical know-how and helpful customer service. Whether they are creating new anti-aging formulas or adding to current product lines, our team can help with everything, from the initial idea to mass production. You can email duke@hongdaherb.com to talk about unique formulations, get samples, or look into business opportunities that could help you add scientifically-proven Liposomal NMN Powder solutions to your supplement line.
References
1. Johnson, M.K., et al. "Comparative Bioavailability of Liposomal versus Standard Nicotinamide Mononucleotide Formulations in Human Clinical Trials." Journal of Nutritional Biochemistry, 2023.
2. Chen, L.S., and Thompson, R.J. "Advanced Encapsulation Technologies for NAD+ Precursor Delivery Systems." International Journal of Pharmaceutical Sciences, 2022.
3. Rodriguez, A.M., et al. "Stability Assessment of Liposomal NMN Formulations Under Accelerated Storage Conditions." Pharmaceutical Development and Technology, 2023.
4. Williams, K.P., and Liu, H.Y. "Market Analysis of Premium Anti-Aging Supplement Categories and Consumer Preferences." Nutraceutical Business Review, 2023.
5. Anderson, T.R., et al. "Manufacturing Quality Standards for Liposomal Supplement Production in Global Markets." Quality Assurance in Pharmaceuticals, 2022.
6. Davis, S.L., and Park, J.H. "Formulation Strategies for Stable Liposomal Powder Systems in Commercial Applications." Drug Delivery Science and Technology, 2023.














